Overview
Electrification of the chemical industry depends on electrochemical reactors that operate selectively and stably at industrially relevant current densities. Our group addresses this challenge through an integrated workflow that links polymer design to device performance. Using organic synthesis as a foundational tool, we design and synthesize functional polymers, including ion-exchange membranes and catalyst binders, and apply them in electrochemical reactors that convert small molecules into fuels and value-added chemicals using renewable electricity. In situ characterization with vibrational spectroscopy probes the polymer–catalyst interface under operating conditions, and long-term stability testing reveals the degradation pathways that must be addressed for practical deployment. Insights from each stage feed back into the next generation of materials, building structure–property–performance relationships that guide rational polymer design across electrochemical energy conversion systems.
Keywords: ionomers; ion-exchange membranes; electrocatalysts, zero-gap electrolyzers; in situ spectroscopy; transport within interfaces; reaction mechanisms.
Ionomeric Binders and Ion-exchange Membranes
An ion-conducting polymer (ionomer) is a polymer in which a small proportion of the constitutional units contains ionic functional groups. Ionomers can be used to constitute (a) ion-exchange membranes and (b) catalyst binders that are added to a catalyst layer in a zero-gap reactor. These polymers set the operating window of any electrochemical application, whether CO2 reduction, water electrolysis, fuel cells, or nitrate reduction to ammonia. They control how water and ions reach the catalyst, how products leave it, and how long the device survives under load.
Various architectures of zero-gap electrolyzers have been developed to achieve industrially relevant energy efficiencies and current densities. A membrane electrode assembly (MEA) stands out among these architectures, in which the cathode gas-diffusion electrode (GDE) is pressed directly onto an ion-exchange membrane without a catholyte. In engineering such devices, it is necessary to employ active, selective, and stable electrocatalysts, as well as highly ion-conducting and chemically stable solid polymer electrolytes.
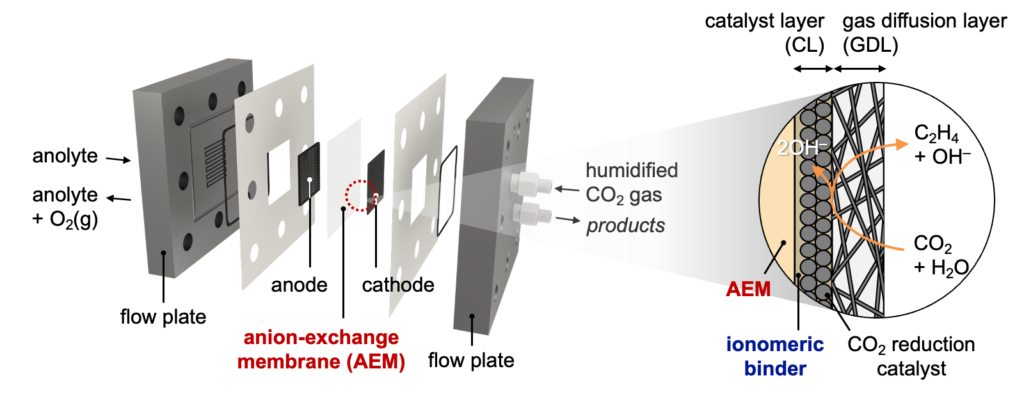
We design and synthesize ionomers for binders and ion-exchange membranes with tunable design parameters, including backbone chemistry, ionic functional group, and ion-exchange capacity. Our research defines these molecular variables as descriptors that link polymer chemistry to electrolyzer performance, thereby providing design principles for next-generation ionomers. This thread runs from monomer synthesis to MEA operation within a single laboratory.
Engineering Catalyst Microenvironments
Electrochemical conversion of small molecules into value-added chemicals is a promising strategy for sustainable chemical production. The success of this approach requires an in-depth understanding of electrocatalysis and the development of efficient electrocatalysts, ion-exchange membranes, electrolytes, and electrolyzers. Product selectivity in a working electrolyzer is set by the local environment around each active site, not by the catalyst alone.
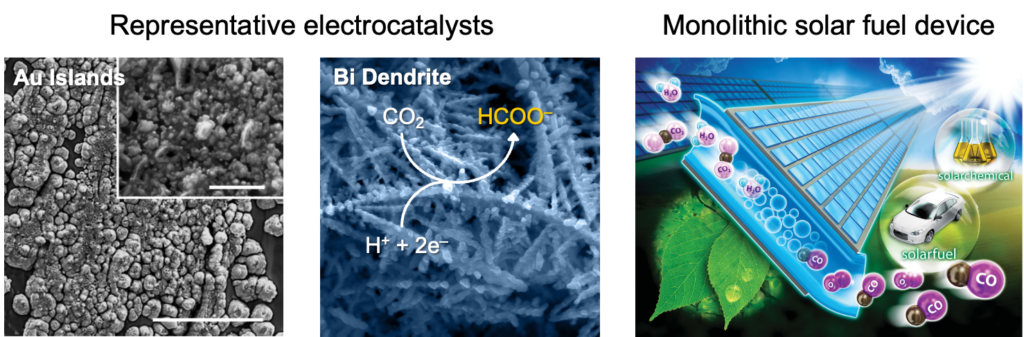
We develop electrocatalysts that produce target products selectively and reproducibly under stable conditions, and we study how the ionomer surrounding active sites shapes a local reaction environment (microenvironment). Side-chain chemistry, charge density, and hydration of the binder modulate the availability of reactants, water, and cations at the catalyst surface, which in turn shifts product distribution and stability. By treating the catalyst and the ionomer as one coupled system, we move beyond catalyst screening toward catalyst-layer engineering. CO2 reduction serves as a representative testbed for this approach, and the same design logic extends to other electrocatalytic reactions of interest.
Device Integration and In Situ Diagnostics
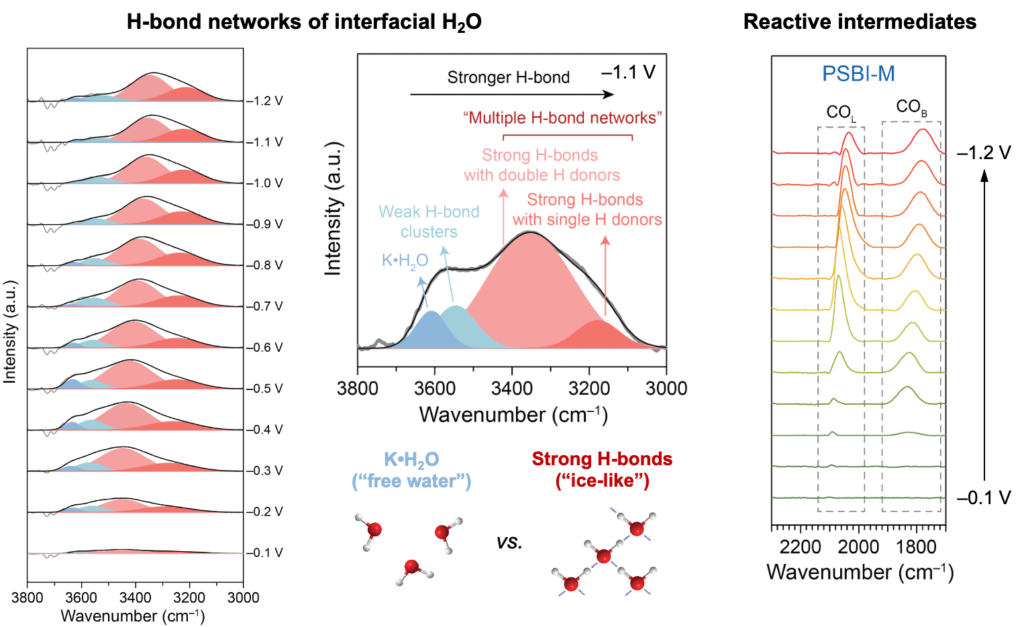
Translating ionomer and catalyst advances into useful devices requires careful engineering of GDEs, MEAs, and the surrounding flow architecture. We operate zero-gap electrolyzers at industrially relevant current densities and use in situ diagnostics to track water management, salt formation, and degradation pathways under realistic conditions. These efforts close the loop with our materials work, since device-level observations feed back into the next generation of ionomers and catalyst layers. The diagnostic and engineering toolkit developed here is not tied to a single reaction and transfers naturally across electrochemical conversion systems.
Platform Applicability
The capabilities developed in our group, including organic synthesis of ion-conducting polymers, catalyst–binder integration, MEA fabrication, and in situ diagnostics, are broadly applicable across electrochemical energy conversion. The underlying science of how polymer chemistry shapes catalyst microenvironments, ion transport, and long-term durability is shared among CO2 reduction, water electrolysis, and other reactions that turn renewable electricity into chemicals and fuels. We pursue fundamental studies with this generality in mind, so that each advance contributes to the broader field rather than to a single application.
